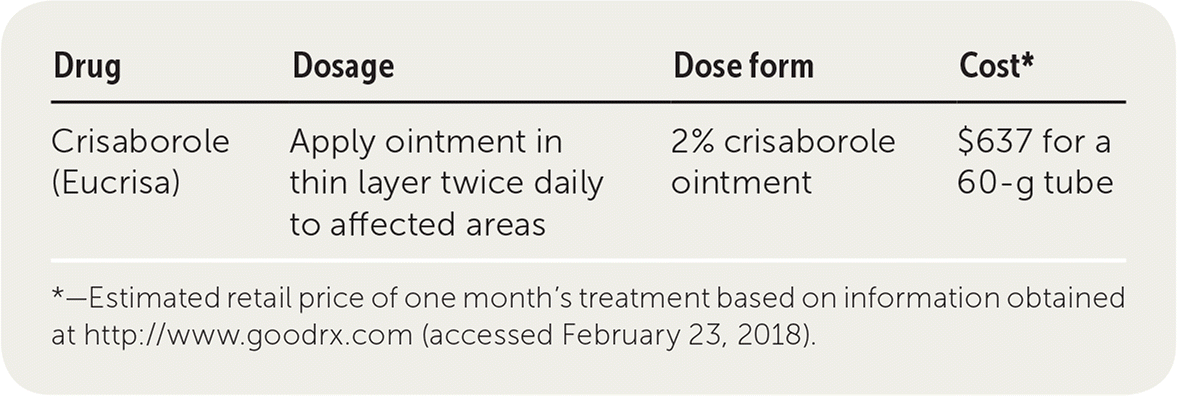Pfizer Eucrisa Ointment Has Been Approved By The US FDA For Use In Infants And Children Aged ≥3 Months! - Industry news - News - Hefei Home Sunshine Pharmaceutical Technology Co., Ltd

Positive Top-Line Results Seen in Study of Crisaborole for Mild to Moderate AD in Young Children - Practical Dermatology