FDA-Approved Patient Labeling Patient Information CAYSTON® (kay-stun) (aztreonam for inhalation solution) Read this Patient Inf
Getting to know CAYSTON What is CAYSTON? Who should not take CAYSTON? What you should tell your doctor before taking CAYSTON How
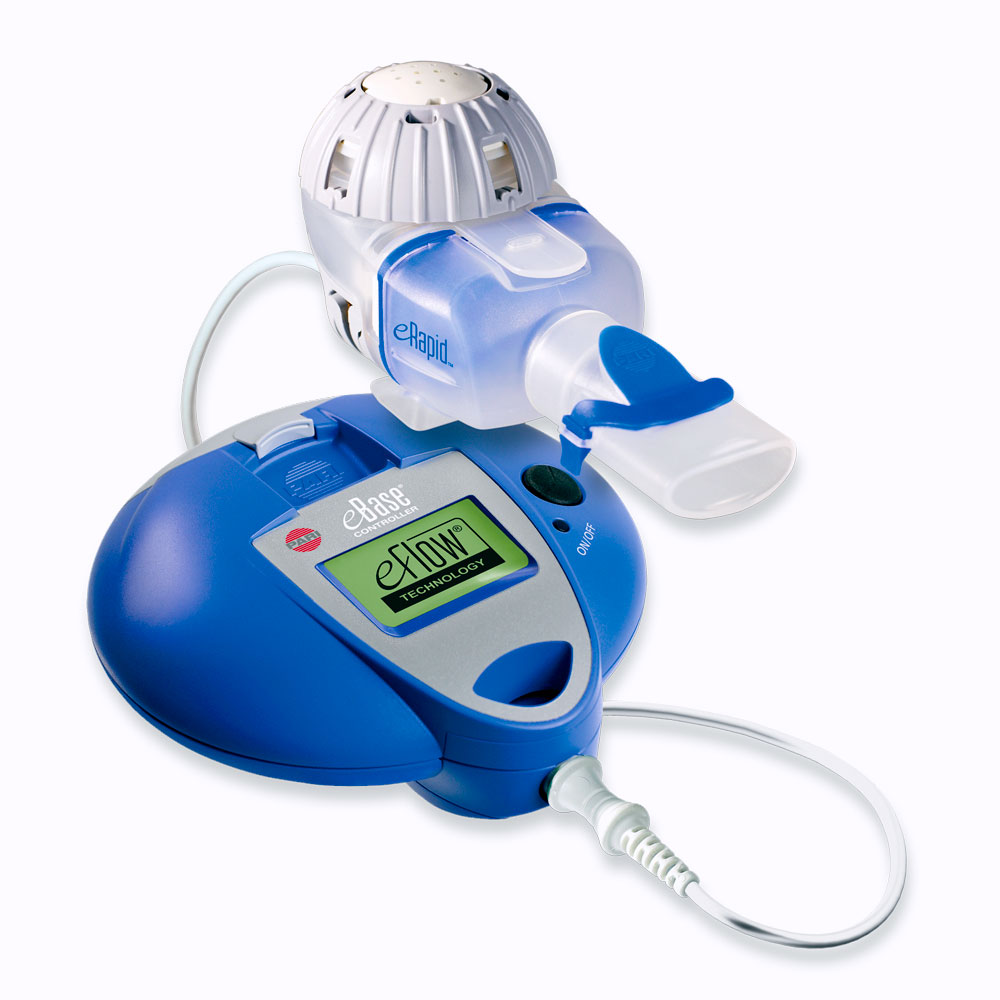
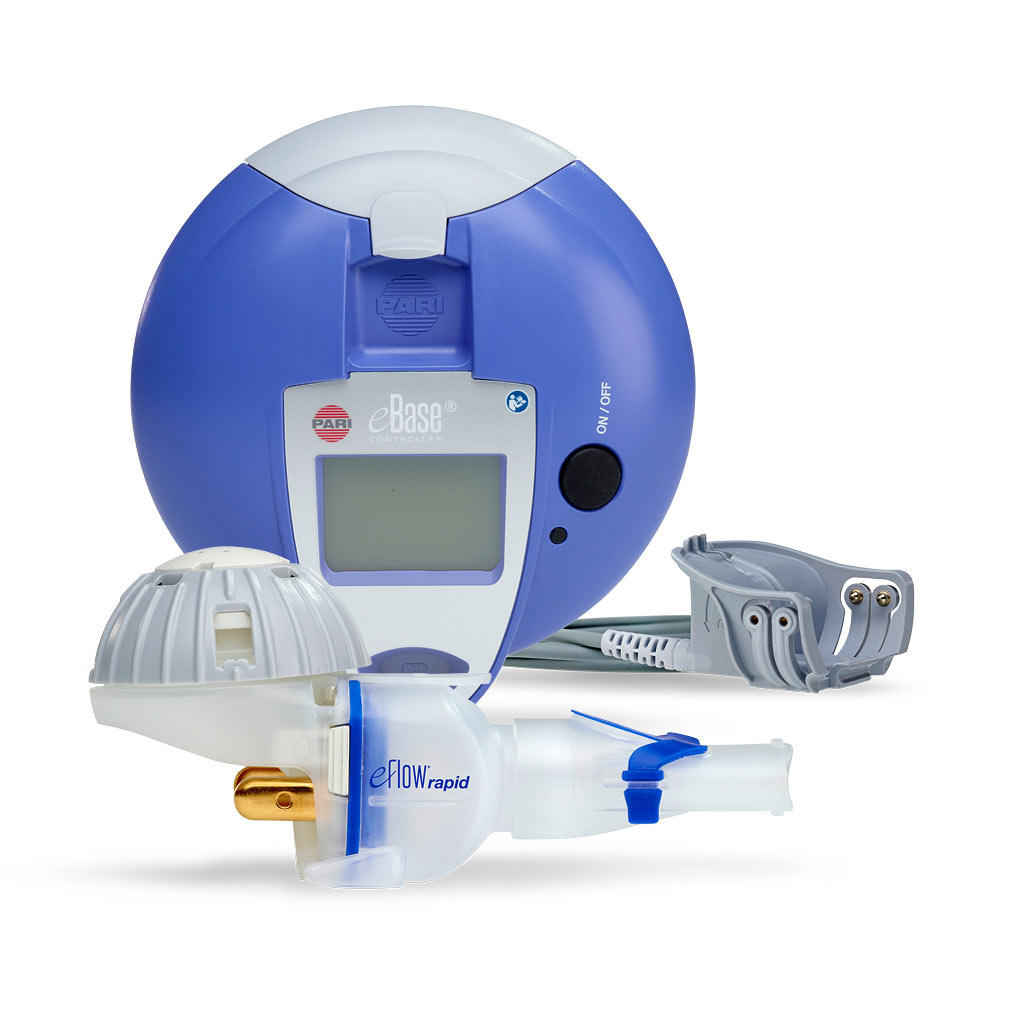
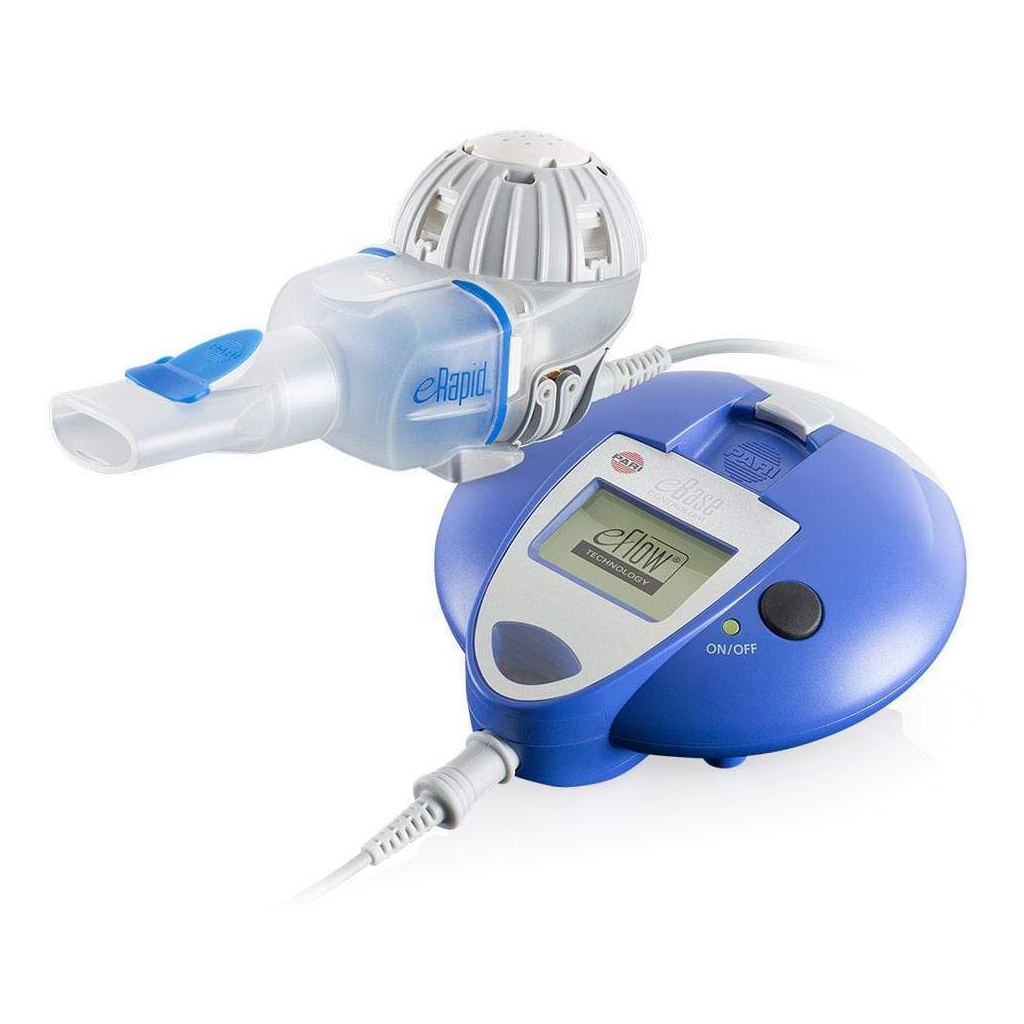
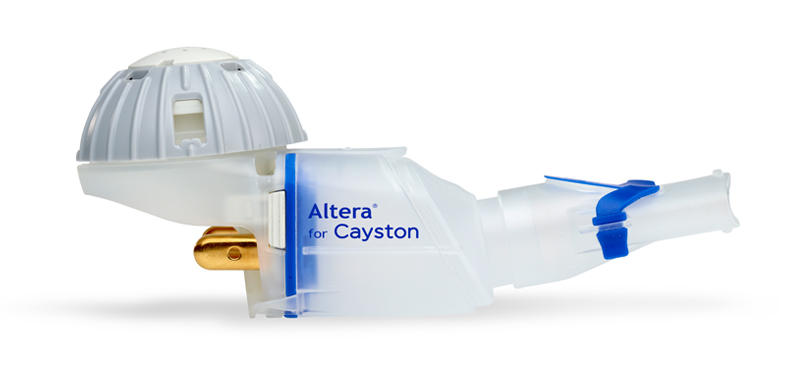
Guidance on the Use of eFlow Nebulizers (Altera® and Trio®) April 6, 2010 David E. Geller, MD and Kenneth C. Kesser, RRT Nemou
Getting to know CAYSTON What is CAYSTON? Who should not take CAYSTON? What you should tell your doctor before taking CAYSTON How